The antimicrobial potential of bacteriophages – UK Parliament Inquiry 2023

Ruby CY Lin, Ameneh Khatami, Anton Peleg, Stephen Stick, Steven Tong, Morgyn Warner, Stephanie Lynch and Jonathan R Iredell*
* corresponding author
The UK Parliament Science and Technology Committee opened an Inquiry on the “antimicrobial potential of bacteriophages”.
In total, there was one oral evidence transcript and 34 articles of written evidence submitted from scientists and health practitioners based at universities, healthcare providers and commercial entities within UK and internationally.
Most submissions highlighted the lack of regulatory clarity and limitation in production capacity in UK, as well as international jurisdictions’ different approaches to regulate, develop and implement phage-based therapies. All agreed that 1., antimicrobial resistance (AMR) is a critical global issue, 2., all are committed in tackling AMR, and 3., phage therapy is safe and effective against AMR when used appropriately.
Within the U.K., and indeed international jurisdictions such as Australia and the U.S., it was highlighted that lack of familiarity with phage therapy is a barrier to implementing such into clinical practice. Clinical microbiologists and scientists have highlighted several questions that remain to be elucidated, namely:
- Phage specificity: one phage to one bacterium or phage cocktails. Does this determine phage production pipelines? Is treating phage production similar to an antibiotic production pipeline sustainable?
- Phage engineering: lack of lytic phage against some bacteria leads to the need for training phages or genetically modifying (GMO) phages. Does this need more safety data or do we implement Good Lab Practice during production of such GMO phages? Is Good Manufacturing Practice the solution? We have highlighted the Belgian Magistral Model[^1] as well as the current practice in Australia[^2].
- Bacterial host engineering: lab strain bacterial hosts and/or patient-derived bacterial hosts are currently used in phage production. Presence of virulence and AMR associated genes in such strains as well as the therapeutic phage are highlighted through genomic sequencing. Can we engineer a bacterial host that can upscale a family of phages? This is not only a production question but a science question.
- Bacterial resistance: bacteria and phages co-evolve. The dynamic interaction is well known but not well studied. During phage therapy, the interactions may impact how treating clinician/s administer phages over a timeframe. Real-time monitoring enables clinicians to make better informed decisions [^2].
- Immune response: anti-phage antibodies are known to neutralise phages in cell culture and in animal experiments but there are limited data demonstrating human immune responses in vivo [^3] to this observation. There is a clear need for in vivo data informing phage kinetics and the human host response. Dynamic interactions between phage, bacteria and the human host during treatment can be usefully monitored in real time.
- Treatment time frame: Treatment durations are arbitrarily set at present but mimic typical durations of antibiotic therapy. The development of neutralising antibodies at 7-1 0 days and the amplification effects observed during therapy are potential guides.
Phage Australia Response
On behalf of Phage Australia, Jon Iredell, Ameneh Khatami, Ruby Lin, Anton Peleg, Stephen Stick, Steven Tong and Morgyn Warner, submitted evidence based on our Australian experience in phage therapy, which are described here.
Realising the antimicrobial potential of bacteriophages
In regard to the call for evidence, Phage Australia addressed several of the key questions raised, on behalf of the leadership of Phage Australia.
Several questions were identified in the call;
- How well established is the evidence base for phages as an antimicrobial for humans?
- What are their strengths and weaknesses?
- What regulatory approaches have been used by other countries for the use of phages and what lessons can the UK learn?
- What opportunities does the UK have for regulatory divergence from the EU on phages, and what would the implications be?
- What are the major barriers and opportunities relating to the development and deployment of phages in the UK?
- How well developed is the UK’s phage research and clinical trial pipeline and how could it be improved?
- To what extent is the UK Government ensuring that phage research and development is adequately funded and supported?
The following submission addresses primarily the first two questions.
Executive Summary
The evidence base for phages as an antimicrobial is strong in regard to safety, assuming proper preparation of therapeutic material, but is weak in regard to efficacy in that high quality comparative data from human therapeutic trials are still lacking.
The strengths of bacteriophages are their specificity (thereby avoiding collateral damage to the bacterial ecosystems that protect us), their capacity for in vivo adaptation to bacterial resistance/ avoidance and their potential for ‘autodosing’ (that is, ongoing replication until target bacteria are consumed), their capacity to be used as delivery systems (carriers of antibiotics, small peptides, diagnostic markers and the like), their common additive or synergistic action with antibiotics and their continued activity in the face of antibiotic resistance.
Challenges include the need to have many phages to deal with bacterial diversity (and the diagnostics to quickly manage this), the uncertainty in regard to best-use applications of phage therapy and in regard to regulatory frameworks and finally, their status as ubiquitous naturally occurring viruses that are easily sourced and amplified (which creates questions in regard to IP security). These latter three in particular have a chilling effect on venture capital investment but all these aspects can be effectively managed.
Regulatory pathways are unresolved and are being worked through in Australia in an attempt to provide both export-ready (GMP track) and ‘compassionate use’ (GMP exempt) pathways.
The UK government has an opportunity to establish national integration of science and medicine in the service of public health and into a huge range of other areas in which bacteria have potential to cause problems (e.g., agriculture, aquaculture, food production, industrial biofouling and biocontamination). This will build important sovereign capacity in health and biotechnology, create smart jobs for the nation, and establish an international leadership position which is unlikely to be challenged in the immediate future as no other governments appear to have taken such as step.
It is likely that industry will continue to regard the regulatory uncertainties and the other risks outlined above, as poor venture capital investment. Government-led initiatives are uniquely placed to de-risk the commercial investments ultimately needed to move phage therapy into the mainstream pharmacopoeia. Government has unique regard to sovereign risk and public good benefits from improved care in public hospitals, and extensions into other non-health endeavours.
GMP pathways
GMP pathways need to accommodate the diversity of phages required. Careful linkage with surveillance programs and a tiered approach to working cell banks in GMP pathways can be used to develop an economically viable model.
GMP exempt pathways
A discussion paper for GMP exempt strategies is attached (see over). This does NOT represent the position of the Australian government.
Clinical training and clinical networks
In order to ensure that phage therapy is used most appropriately, it may be necessary to weigh the benefits of initially restricting access to phage therapy to an expert sector of clinical prescribers (e.g., Infectious Diseases physicians) in the same way that new anticancer drugs would not be widely available for non-experts to use. A training course might be developed to enable graduates to access therapeutic phages, in the way that HIV therapy was made available to non-expert prescribers in the Australian system through the “S100 scheme”.
An important early step taken in Australia has been to develop consensus around the need and the initial approach to monitoring, so that emergency IND (as per U.S.) or ‘compassionate use’ indications can be done in the best possible way and to yield the most useful data. This approach is exemplified in the consensus protocol generated by the Phage Australia clinical network.
Contents
1. Principles
- Endpoint QC rather than process-controlled GMP
- Standardised descriptions
- Phage safety
- Production strain safety
- Production safety
- Phage-bacterial matching (diagnostics)
2. Preamble
Bacteriophages are used as a treatment of last resort for patients at risk from severe infections. Since 2007, patients who meet the requirements of the Therapeutic Goods Administration (TGA) Special Access Scheme have had access to phages if they can be provided safely.
The unregulated product, if deemed safe by the organisations and individuals with specific duty of care, has been administered by a variety of routes: topical, intravesical, intranasal, endobronchial, aerosolised and intravenous. Natural and genetically modified phages have been administered individually and in combination with other phages and with antibiotics in severely ill adults and children in hospital and in the home setting.
There is an agreed national protocol for standardisation of therapy and monitoring.
Permission for detailed monitoring has been conducted under the auspices of the STAMP protocol (ACTRN12621001526864). At this time, there are several sites in metropolitan and rural centres with site specific governance to proceed with monitoring.
Dozens of metropolitan and rural sites are now registered for access. National survey has shown that prescribers want structure, monitoring, and oversight. The actual experience with human phage therapy to date is almost exclusively at Westmead but phage therapy units are now developing in other sites (most notably Adelaide, Melbourne, and Perth).
The principal bottleneck is manufacturing.
Most phages for intravenous and nebulised use have been sourced externally from companies (e.g., Armata and APT (Adaptive Phage Therapeutics)) and academic institutions (e.g., University of Pittsburgh) but three Australian patients have now been treated with phages manufactured at Westmead. Four intravenous and three topical phage preparations have been used to date, treating E. coli and P. aeruginosa infections in adults and children.
Permission to use non-GMP product has been sought in all instances from Western Sydney Local Health District (WSLHD) and Sydney Children’s Hospitals Network, whose drug committees have reviewed documents provided from the Iredell lab at Westmead (attached). A similar process has been/ is being developed at Royal Adelaide/ Queen Elizabeth Hospitals in SA, Alfred/ Monash in VIC and Telethon Kids/ Perth Childrens Hospital in WA. All cases have bene notified to TGA through the Special Access Scheme.
3. Production-related certification
3.1 Phage matching (diagnostics)
The matching of bacteriophage to bacterial target is done usually in two ways:
- “plaque assay” - a drop of purified virus "is placed on a lawn of target bacteria (and this can be modified by serial dilution in the “efficiency of plating” method, which may be expressed as a fraction or percentage as a surrogate for the activity of the phage).
- Growth inhibition – this is more conventional but the interpretation is complicated by lack of standardisation e.g. in the ratio of virus to bacteria (the ‘multiplicity of infection’, MOI) and in the ‘training’ of phages (and bacteria).
Comment – this (especially. b) is close to the paradigm of growth inhibition in antibiotic testing in routine clinical microbiology labs and can be adapted into such but needs standardisation and clinical validation.
3.2 Phage safety
The identification of a phage as ‘safe’ is usually by exclusion of the capacity to integrate into the bacterial genome, thus both ensuring an obligate lytic outcome of infection and minimising the risk of transmission (by convention, “transduction”) of unwanted genetic material from bacteria.
This can be done manually but is efficiently done by genome sequencing.
3.3 Bacterial production strain/s
Production of a phage for subsequent therapeutic use is done by growth in a receptive bacterial host. Phage specificity means that this is often a pathogenic host. A strain that is likely to co-purify a dangerous toxin might be considered problematic.
This can also be most efficiently excluded by genome sequencing.
3.4 Batch safety (sterility testing)
By convention, the elimination of bacterial impurities is confirmed by showing that bacterial ‘endotoxin’ (measured in endotoxin units, EU) is below the common ‘pyrogenic threshold’ when dosing. Measurement of EU is well standardised and widely used in GMP bioprocessing.
Bacterial contamination and the presence of other pyrogens or undesirable products may be tested in a variety of ways including monocyte activation tests but there is no clear consensus and the potential risk can be assessed by (3), above.
3.5 Comment
In the absence of classic process control as in GMP, there is opportunity to standardise all of these aspects above. At a minimum, we believe (3.2) and (3.4) are essential. (3.1) is expected to fall under NPAAC/NATA processes and (3.3) is difficult to standardise or regulate but may offer additional confidence when assessing batch safety (3.4).
4. Production processes
4.1 Phage culture and harvesting
Most (not all) bacterial hosts are suitable for liquid culture. This may be as single inoculation or continuous culture (e.g., chemostat) and phages are typically used to inoculate early to mid-log phase cultures. There are advantages to each (continuous vs discontinuous culture) approach.
Some bacterial hosts (such as the non-tuberculous mycobacteria, for which phages are frequently requested) are best harvested from plate cultures of an infected bacterial lawn. This often generates higher yield for other bacteria which can also be inoculated in liquid culture but is also more labour intensive as a rule.
To ensure that all types of bacteria can be most effectively targeted by phage, all bacterial growth conditions must be accommodated in a regulatory framework.
4.2 Phage purification
Physical separation: Initial harvest (‘lysates’) contain bacterial debris and live bacteria. The first step is usually centrifugation prior to filtration. This step may include density gradient separation with Cs salts, for example.
Chemical separation/ chromatography: Semi-purified lysates after centrifugation/ filtration steps are often subjected to differential binding columns and resins, sometimes with specialised properties, to dissociate attached/associated bacterial membranes/ debris. This may involve salts, detergents and/or organic solvents which then have to be further cleaned by filtration/ buffer exchanges.
These simple physicochemical methods have been used widely for decades but are labour intensive.
Newer approaches to chromatography lend themselves to automation and are increasingly popular because of their ease of use and potential for standardisation.
4.3 Excipients and additives
Simple sugars (e.g., trehalose) are commonly added as stabilising agents in freeze-dried or spray-dried preparations.
4.4 Chemical and genetic modifications
Genetic engineering of phages is commonplace and has been widely deployed in human therapy, including in seriously ill children at Westmead.
Chemical modifications by ligation of antibiotics, biofilm-disrupting compounds, imaging molecules and the like, are relatively simple and can be done in late stages or after completion of purification.
5. Current phage therapy pipeline under STAMP
Current statistics by Phage Australia
- 300+ locally biobanked phages.
- 47 patients in screening stages.
- 23 patients with phage matches (25 patients as of February 2023).
- 14 phages produced for Australian patients and passed local QC.
- 2 patients treated in 2022 with Australian phages (currently a 3).
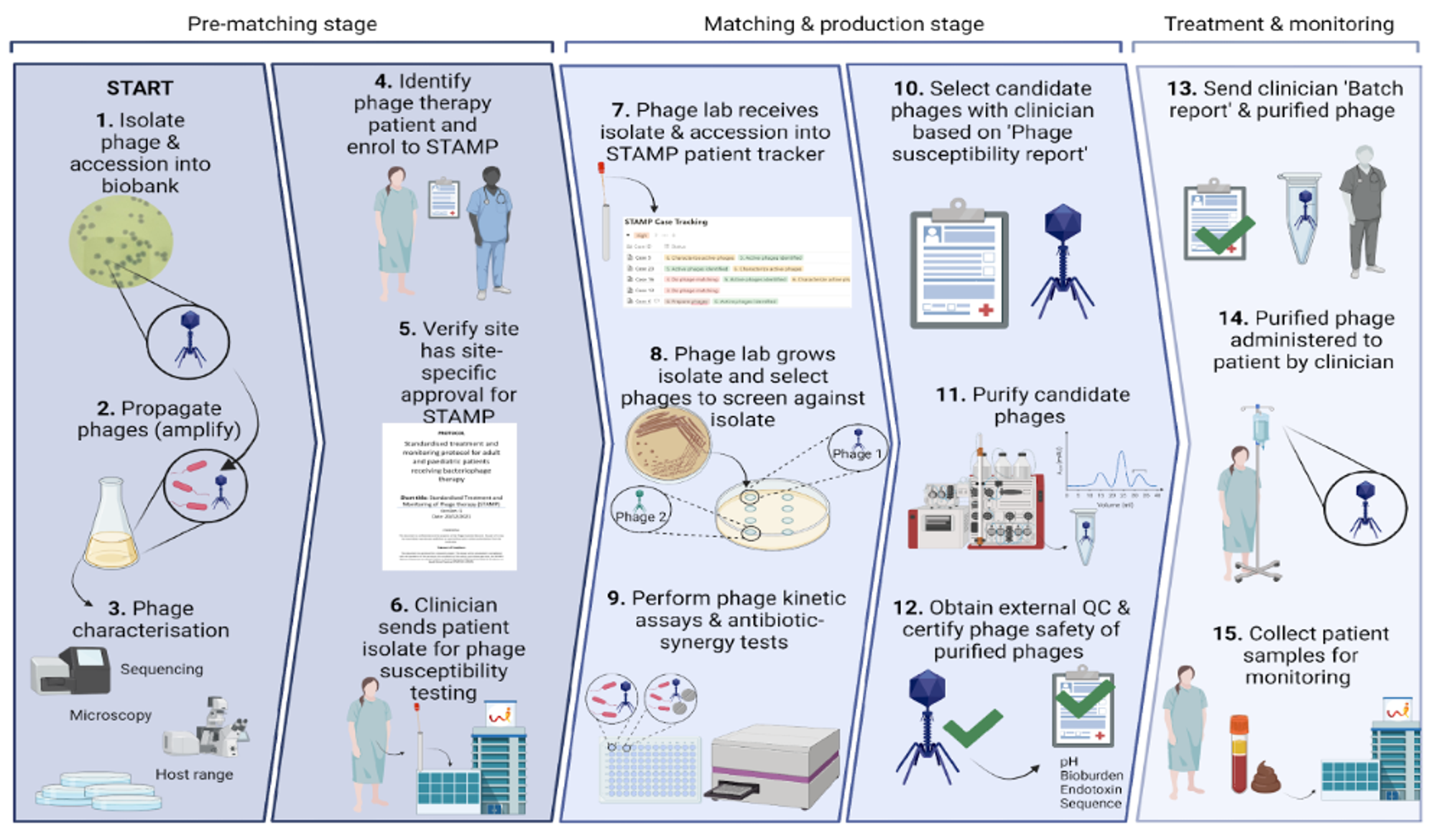
Fig. 1. Phage Australia phage therapy pipeline (Credit: Stephanie Lynch). Multiple phages covering multiple species should be available to be drawn upon. Patients referred are assessed for suitability and a bacterial isolate is sent for phage susceptibility testing. In 2022, there were 47 deidentified patient requests with 56 isolates received in the Iredell lab. If suitable phages are identified (e.g., we identified a potentially suitable phage for 23 patients in 2022), the doctor confirms SAS category eligibility/ status and seeks informed consent to proceed with safety monitoring as per STAMP protocol from the patient and candidate phages then go forward to purification. Safety reports (manufacturing host genome, phage genome, batch quality and safety, diagnostics) are furnished to the local drug committee. If the relevant drug committee authorises and SAS criteria are met, therapy is made available. During phage therapy, we receive patient blood samples for monitoring according to the STAMP protocol.
5.1 A case study
5.1.1 Enrolment of a patient under STAMP
An immune compromised adolescent patient with chronic sinusitis and contiguous osteomyelitis, with recurrent bacteraemia was proposed under category A SAS criteria for experimental phage therapy. A swab from the infection site isolated 4 Pseudomonas isolates which we received one week after being notified of the case on the 30th of September. Suitable phages were identified and purified. See Fig 2.
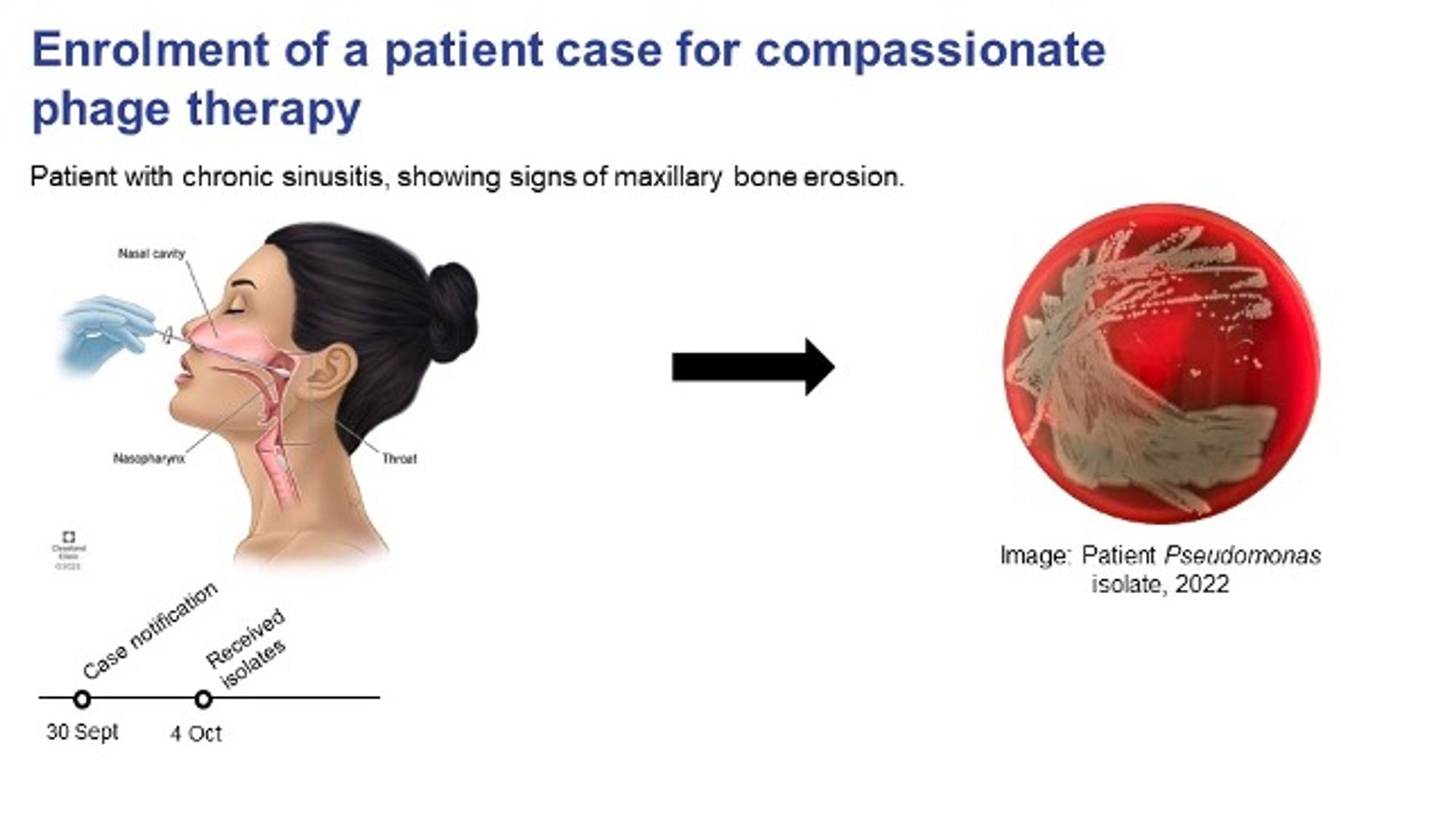
Fig. 2. Case notification and enrolment of patient. Patient matching commences (Credit: Stephanie Lynch).
5.1.2. Phage susceptibility testing on patient bacterial isolate. Spot Test.
The first step of identifying a phage is conducting a spot test/ plaque assay. A bacterial lawn of the patient bacteria is created and 10 µL aliquots of different phages are tested. (Fig 3).
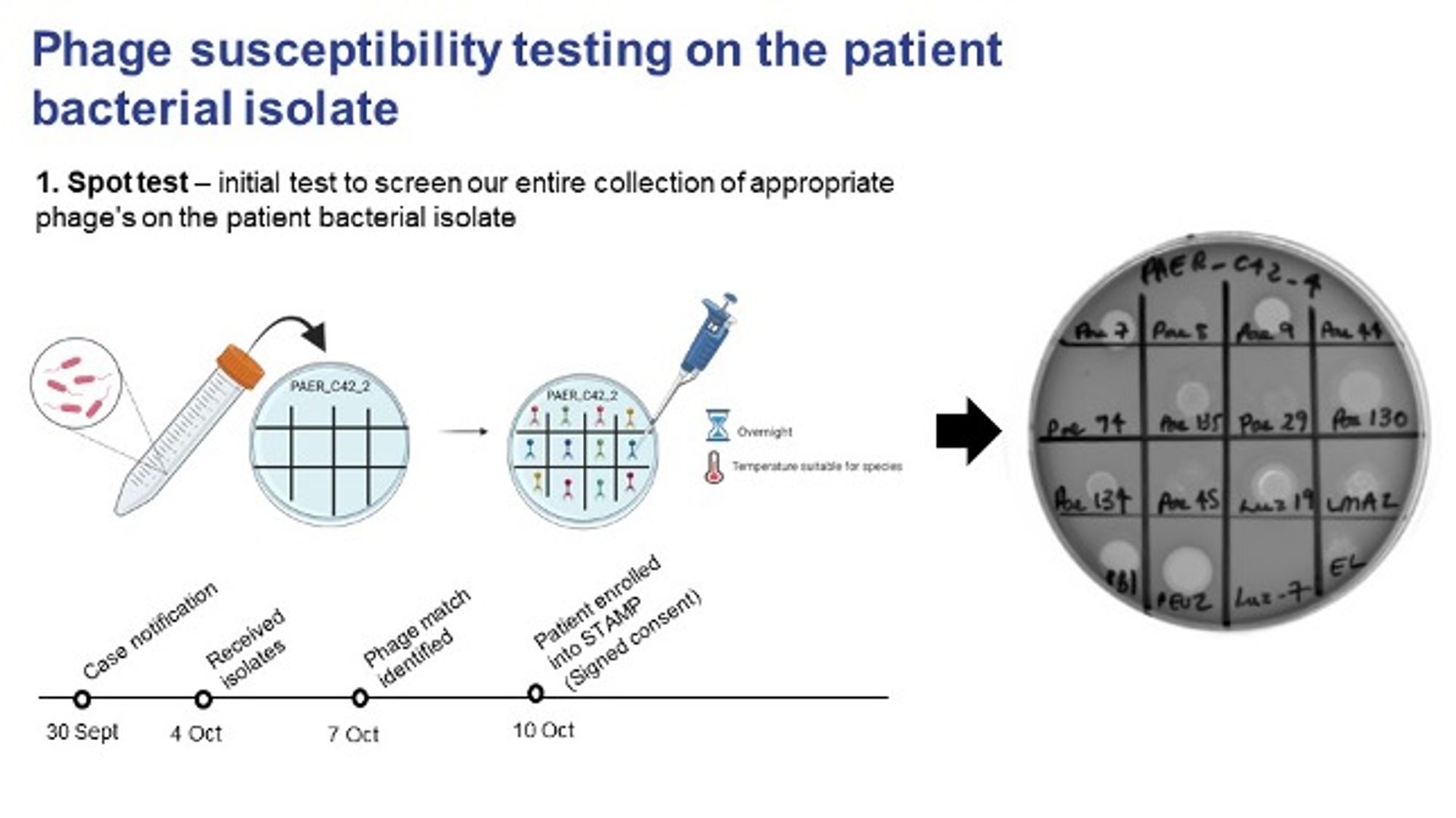
Fig. 3. Phage susceptibility testing (Credit, Stephanie Lynch). The Spot Test (Plaque assay – see 3.1a). revealed candidate phages just three days after receiving the patient isolate, each with varying zones of clearing (lysis).
5.1.3. Phage susceptibility testing on patient bacterial isolate. Efficiency of Plating test.
Serial dilution provides a semi-quantitative phage titre, with a reference bacterial host isolate as a control to compare activity vs the patient isolate. Fig. 4.
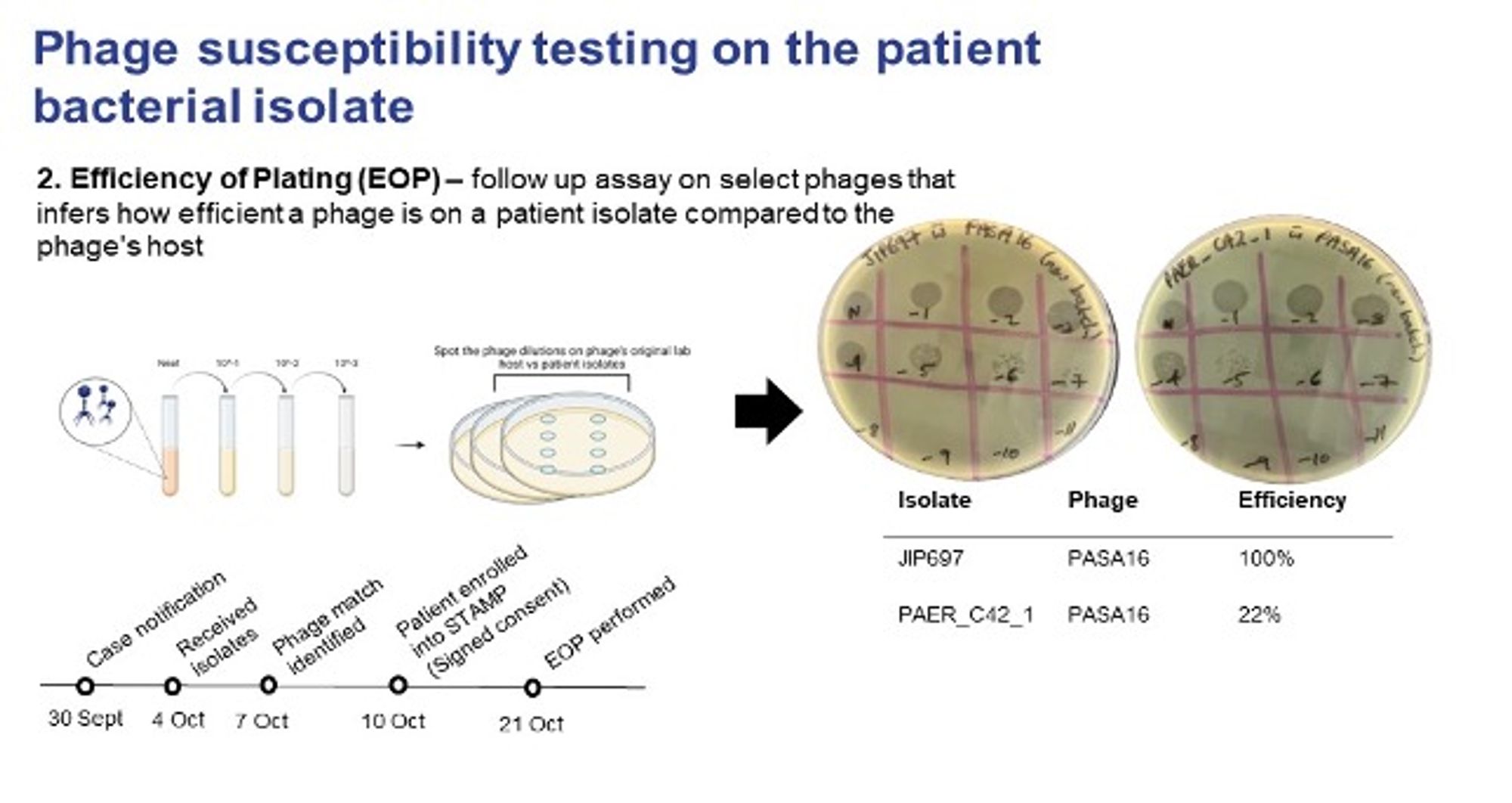
Fig. 4. Phage susceptibility testing. Efficiency of plating test (Credit, Stephanie Lynch).
5.1.4. Phage susceptibility testing. Growth assay (3.1.b).
The growth (inhibition) assay tests growth of the patient bacterial isolate in broth in the presence of phage or phage combinations over an 18-hour period. If the phage is effective in vitro, we see significant inhibition of bacterial growth.
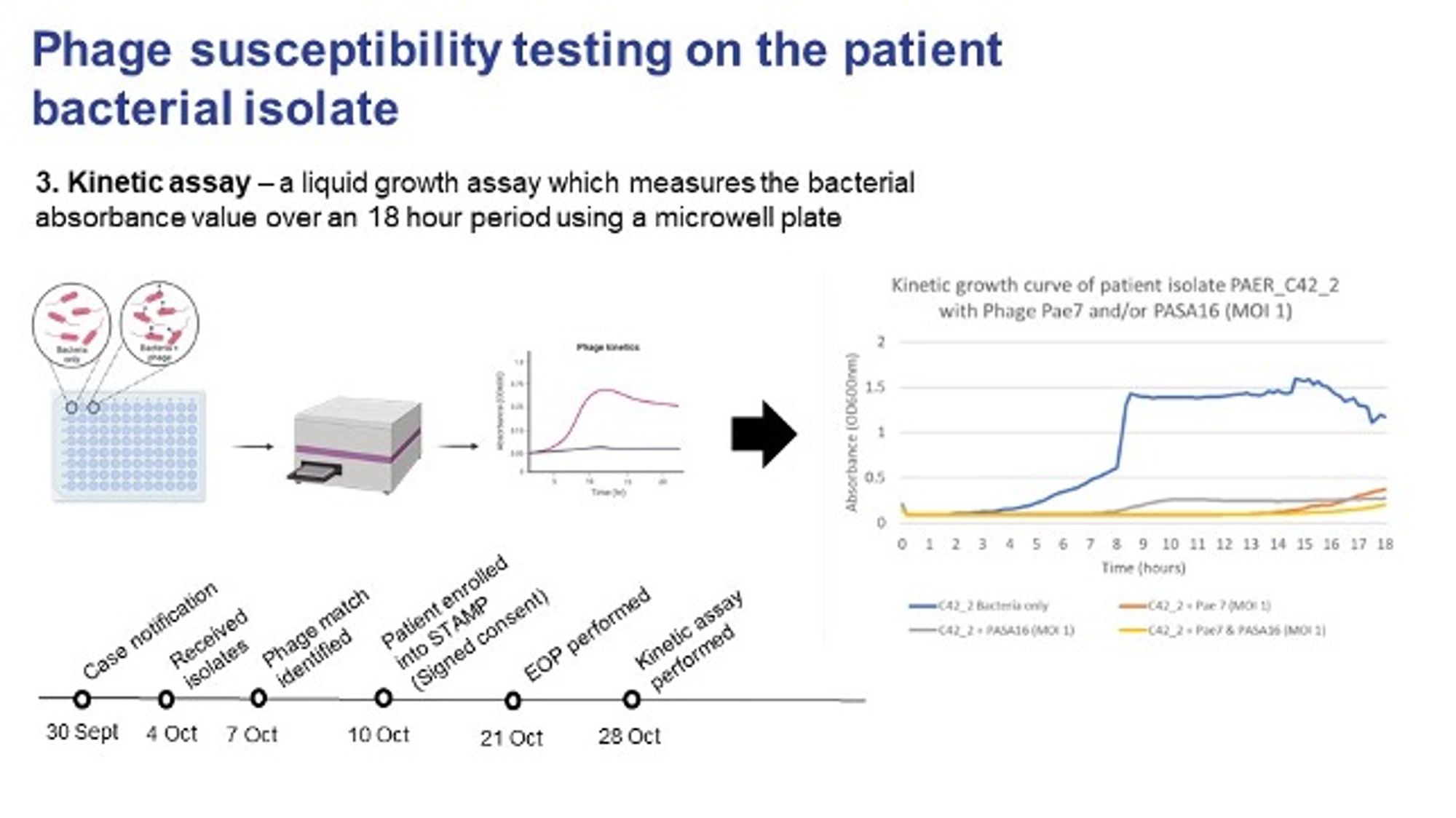
Fig. 5. Growth (kinetics) assay (Credit, Stephanie Lynch). The line in blue indicates the normal growth curve of the patient isolate with no phage added, then the grey and the orange lines show the bacterial growth curves in the presence of phage, the two colours indicating different phages. The yellow line is the growth in the presence of both phages that showed inhibition of growth for 18 hours.
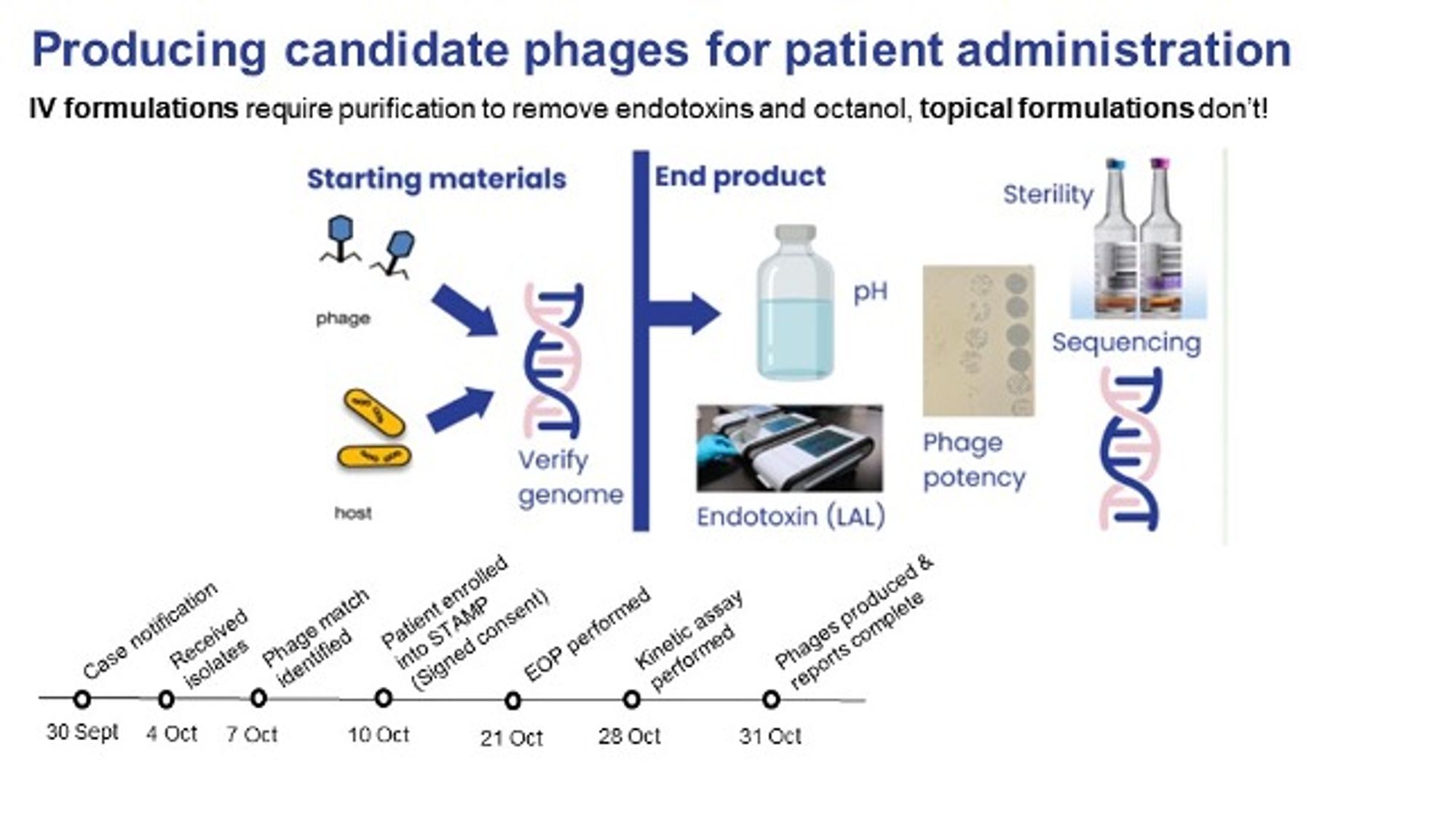
Fig. 6. Production and purification (Credit, Stephanie Lynch). Phages replicate by bursting the bacteria and so the product requires purification to remove bacterial debris such as endotoxin. The final phage preparation is tested for pH, sterility, and endotoxin content.
5.1.6. Patient administration and therapeutic monitoring
The STAMP protocol allows for monitoring of any dosing schedule but a default schedule of 1 or 2 doses per day of 1 mL of phage at 1 x 109 phages/mL is a common choice for 14 days, with monitoring at preset timepoints.

Fig. 7. Phage administration and laboratory monitoring protocol for standard 14-day intravenous/oral phage therapy. Adapted from Table 2, Khatami et al., https://bmjopen.bmj.com/content/12/12/e065401
6. Summary
Bacteriophages are not produced under full GMP frameworks anywhere in the world but are widely used when antibiotics fail and have been used for over a century.
The definition of a ‘safe’ phage and a ‘safe’ product has international consensus around;
- the phage’s ability to integrate or to encode toxins or AMR genes, and of
- the product to contain endotoxin that does not exceed the internationally accepted pyrogenic threshold of 5 EU/kg/ dose.
The dose, interval and duration is less certain but twice daily dosing at 10e9 pfu for one or two weeks is a norm that is consistent with antibiotic therapy options and has considerable data to support it in recently published Australian literature [^4].
Standardisation of phage and batch quality control, as described above, is a minimum safety expectation in the absence of a full GMP pathway.
The STAMP[^2] protocol provides standardised monitoring and ensures that prescribers are appropriately networked to the expertise available in the Australian system.
January 2023
Download a PDF of this White Paper
[^1]: Pirnay JP, Verbeken G, Ceyssens PJ, Huys I, De Vos D, Ameloot C, Fauconnier A. The Magistral Phage. Viruses. 2018 Feb 6;10(2):64. doi: 10.3390/v10020064.
[^2]: Khatami A, Foley DA, Warner MS for the Phage Australia Clinical Network, et al. Standardised treatment and monitoring protocol to assess safety and tolerability of bacteriophage therapy for adult and paediatric patients (STAMP study): protocol for an open-label, single-arm trial BMJ Open 2022;12:e065401. 1
[^3]: PFA, Lin RCY et al., Safety of bacteriophage therapy in severe Staphylococcus aureus infection. Nat Microbiol. 2020 Mar;5(3):465-472. doi: 10.1038/s41564-019-0634-z; And Khatami A, Lin RCY et al., Bacterial lysis, autophagy and innate immune responses during adjunctive phage therapy in a child. EMBO Mol Med. 2021 Sep 7;13(9):e13936.
[^4]: https://www.nature.com/articles/s41564-019-0634-z; https://www.embopress.org/doi/full/10.15252/emmm.202113936
To cite this white paper:
Lin, RCY et al. Westmead Institute for Medical Research (2022). The antimicrobial potential of bacteriophages – UK Parliament Inquiry 2023. Retrieved from https://phageaustralia.org/blog/antimicrobial-potential-of-phages
NB: Please feel free to write to me with your thoughts. I extend a challenge out to entrepreneurs and intrapreneurs to help us refine business plans and business models.